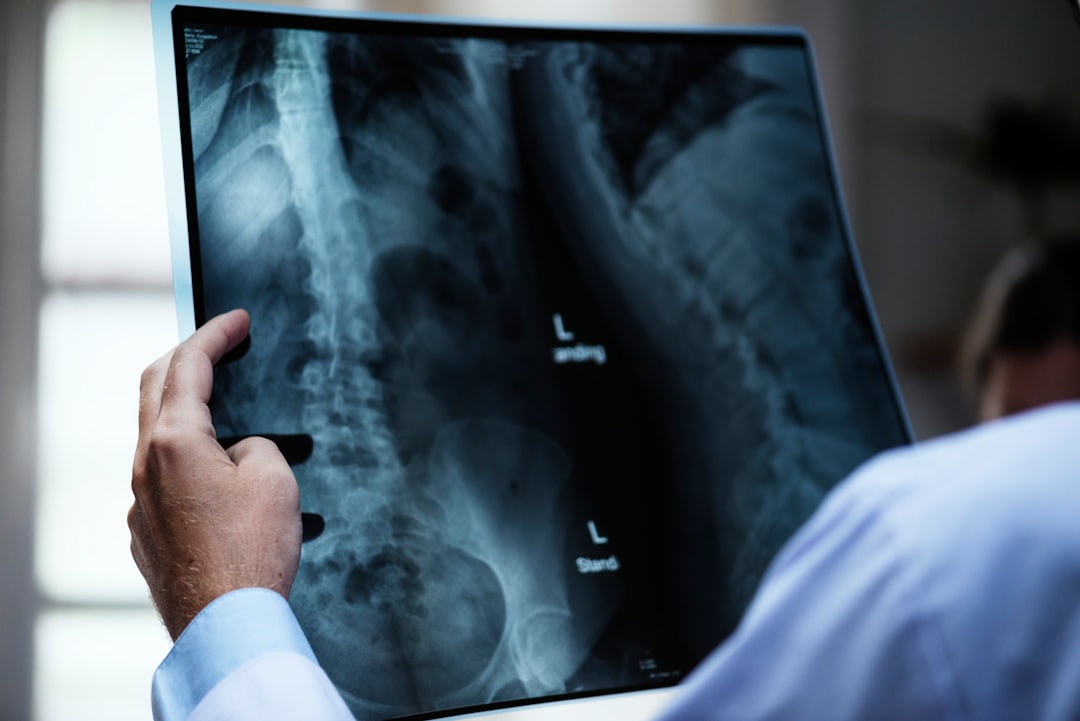
If you take part in a clinical trial, you may get tests or treatments in a hospital, clinic, or doctor's office. Participants in a clinical trial are often treated in the same way as other patients who aren’t in a clinical trial, but are more carefully monitored and may have more tests done.
Clinical trials take place in doctors’ offices, medical centers, community hospitals and clinics, and veterans’ hospitals around the world. A particular trial may be offered at only one location, but some are conducted at many different hospitals across the world.

"To my surprise the entire experience felt no different from any other hospitalization, or procedure; honestly, it was probably the best care I have ever received in my many years as a patient!"
Chanel White, a patient advocate and scleroderma clinical trial participant
Once you go through the process of finding a clinical trial you can participate in and the informed consent process for a study, here's what you can expect from participating in a trial.
Additional medical tests and monitoring
One of the main differences between receiving treatment as part of a clinical trial versus standard care is that there might be more medical tests and attention from medical staff as part of a clinical trial.
Before starting the new treatment, members of the study team will usually look through your full medical history. Depending on the trial, you may need to have blood tests, imaging tests, and/or a physical exam done if you haven’t had them recently. These are needed to be sure that you meet the requirements to participate, and to help ensure your safety.
You may also have tests done more often to find out how well the treatment is going, and check on how you’re doing. You’ll likely get more attention as a participant in a trial than you would otherwise. The doctors and nurses may examine you more often and will want to know if you’re having any side effects (also known as adverse events) while being treated.
Because the possible side effects may not fully be established, it’s very important to let the research team know how you're feeling. They can use this information to decide if symptoms you’re having are related to the clinical trial, if you need any special attention, or your treatment needs to be changed.
Safety of participants is strongly regulated by many organizations, and it's important to remember that you can always leave a study at any time, for any reason.
Keeping a health journal during the trial
While participating in a clinical trial, you may be asked to write a health journal to record your symptoms, diet, and everyday life. This information can help determine common side effects of the treatment.
If you’re required to keep a health journal during the study, what you need to include will be outlined for you. For example, you may be asked to record side effects and symptoms in more detail than your daily activities. Ask about their expectations for patient journals to make sure you’re recording the right details.
Even if your study isn’t requesting a health journal, having a health journal can help you keep track of your health. Some patients find it helpful to keep a digital health journal on their phone or mobile device. This can speed up the journaling process and also allows you to easily share information with your doctors and nurses.
We recommend choosing a consistent place to take notes, written or typed, that can travel easily or be accessed remotely at your appointments.
What to include in your journal
While each patient’s journal looks a little different, entries usually address similar topics. Recording the date of the entry will help you keep track of patterns in your health. Here is a list of some items you might want to include in your journal:
-
Details about each appointment (information received, lab work done, procedures and subsequent reactions etc...)
-
Name and dose of prescribed drugs, especially if it is information that is not immediately available through a paper prescription
-
A running list of questions, concerns, and comments (what's working, what isn't, etc...)
-
Time(s) of the day you take your medicine
-
Diet and timing of meals
-
Daily activities and exercise
-
Symptom progression
-
New or recurring side effects
-
Changes in mood
Preparing to participate in a trial
There are a number of things that you can start to keep in mind, even before you start participating in a study. Planning around insurance coverage, travel and lodging logistics, and the general schedule changes that come with participation can help you feel ready and will ensure that your entire process runs smoothly.
Your team of researchers and the clinical trial coordinator (in addition to your primary care doctor, your friends, and your family), can all help support you throughout your clinical trial journey to make things easier.
Telling others you're participating
While participating in your trial, you may want to discuss your trial with your friends, family, and even coworkers. Here’s some advice on how to navigate these conversations.
Talking to friends and family
Discussing your trial with family and friends can be an important step in your journey, as they can provide a strong emotional support system. Letting them know your needs, and ways they can best support you can ensure that you both feel more comfortable.
When you first tell your friends and family about your trial, they’ll probably have a lot of questions. Consider preparing your responses to the following commonly asked questions:
-
Why are you participating in a trial?
-
Will there be any side effects or risks in the trial?
-
How long will the trial last?
-
How can I help you throughout your trial?
It’s natural for your loved ones to have questions or concerns about the study. Helping them understand the ins and outs of how the FDA, Institutional Review Boards, and trial coordinators ensure patient safety can be a great place to start this conversation.
Finally, some patients also find it helpful to bring a family member or friend to trial appointments. This is just one more way to feel supported through your trial and can make discussing your trial easier.
Informing your employer
First, it’s important to note that you are not obligated to inform your employer. Some patients use vacation days to participate in their trial, and don't find it necessary to inform their workplace. Still, you may decide to inform your employer should you need to request time off from work.
If you decide to inform your employer, let them know that you are enrolling in a clinical trial and may require time off from work. Once you receive your official trial schedule, you'll want to include the start date, end date, and frequency of appointments of your trial, if possible. Also, make sure to review your company’s medical leave policy to determine the pay status and availability of medical leave.
You may find it necessary to outline your reasoning for enrolling in a trial, such as advancing treatment options for others with your condition. It’s best to notify your workplace sooner, rather than later to make coordinating time off from work easier.
Managing your travel and lodging
Before enrolling, you should be notified if and how your trial plans to compensate its participants. Make sure to discuss all available travel compensation, because this can be one of the most expensive parts of trial participation. All the different amounts of travel compensation should be included in your Informed Consent Form. Once this form is signed, it is very difficult for researchers to change the terms of compensation, even if there was a mistake.
Travel
To get to your clinical trial, you’ll likely have to make travel arrangements for your appointments. Whether that’s driving yourself, taking a taxi, or flying, planning your travel to a clinical trial can be difficult. Here’s some advice for when you're trying to make those plans.
Common forms of reimbursement
Here are some ways researchers compensate for travel:
-
Prepaid gas cards
-
Parking vouchers
-
Taxi and ride sharing vouchers
-
Airfare credits
It's important to note that not all trials cover patient travel expenses - and even the ones that do may not fully cover your expenses. That’s why it is important to save all necessary receipts for travel costs. Without these, you may not receive reimbursement. Also, medical-related travel is tax deductible, which is another reason you’ll want to keep track of your receipts.
Overnight stays
For overnight clinical trials, you’ll also have to consider where you'll be staying. For some trials, you are able to sleep overnight at the trial site, but for others, you will likely need to stay in a hotel or Airbnb.
Like travel expenses, lodging expenses are sometimes covered by trial sponsors. If this is the case, you may have to pay for your hotel room out of pocket and then be reimbursed for the charges later. Other trials will pay for the room in advance, and you won’t need to get reimbursed.
Even if your lodging expenses are not covered, make sure to save your receipts. As with travel, lodging expenses for medical care are tax deductible.
When packing for an overnight trial, you’ll want to contact your study site; researchers often provide snacks, music, games, basic toiletries, and other items that you won’t need to pack. Here are some common items to make sure you have on hand:
1. Identification and Medical Forms
To start your trial, you’ll most likely need a government-issued photo ID, such as a driver’s license or passport, to check in. Also, make sure to bring all relevant medical information that you may need.
2. Clothes
In general, you’ll want to bring comfortable clothing that is relatively light and warm. You should pack one outfit per day you'll be at the trial site and one extra outfit just in case you lose any clothing.
Here's a breakdown for each clothing item:
- Shirts: flexible and breathable. Dri-fit shirts are an especially good choice, since they wick sweat and keep you warm.
- Pants: loose-fitting and lightweight. Thin sweatpants and athletic pants should work well.
- Underwear and socks: comfortable and breathable. Comfort is key here, because you may be sitting for long periods of time during your trial visit.
3. Toiletries
If your study site doesn’t provide toiletries or you want to bring your own, don’t forget to pack all liquids in travel containers to save space.
Here's a list of toiletries you may want to pack:
- Shampoo, conditioner & body wash
- Toothbrush & toothpaste
- Comb/brush
- Deodorant
- Razor & shaving cream
- Towel
4. Entertainment
You’ll probably want to bring your phone or tablet for communication and entertainment during your stay at the trial site. Also, consider bringing a laptop if you’ll be at the site for several nights. Finally, think about bringing along a good book for some reading during downtime.
Organizing information
As you go through your trial experience, you will receive a lot of information, and have a busy schedule to keep track of. Planning ahead will help you with keeping track of your progress through the study, and staying on top of your schedule.
We recommend setting up an organizational system before you begin the clinical trial process. It’s helpful to keep all documents in one place (whether they’re electronic or physical), with notes, and a calendar of important dates.
Here’s an outline of useful information to organize, and tips on how to go about doing it.
Documents to keep on file
These are all the things you should have a physical and/or electronic copy of:
-
Treatment plans
-
Prescriptions
-
The most current copy of your medical insurance coverage
-
Insurance claims filed by you, your doctor, or your hospital
-
Explanation of benefits (EOBs) statements from your insurer
-
Insurance reimbursements and claim rejections
-
Appeal letters
-
Check and credit card receipts
Dates to keep in a calendar
For time specific information, we recommend recording the dates in a calendar format so you can quickly glance at it for reference. Particularly important dates include:
-
Upcoming medical visits and calls
-
Treatments received
-
Communication with insurance (such as the date an appeal was filed)
-
Medication (time of start and finish)
Additional organization tips
1. File your bills with their corresponding statements
Even though you may receive the explanation of benefits (EOB) statement separately from the bill, keeping them together will make cross-checking easy and efficient. To this point, always compare the bill you receive with the EOB statement from your insurance company. Inquire about any differences you might detect between them, and always speak up if you’re concerned your bill is incorrect.
2. Organize in chronological order
This one might seem obvious, however, it is hard to predict the amount of documents you'll end up needing to store. Organizing your records in order of when you received them will allow you to easily find a file if you ever need to reference a certain one.
3. Backup your files
If you choose to store things electronically, make sure you have a secure backup system in place. Whether it's printed hard copies, a flash drive, or the cloud, have a backup plan in case your computer crashes.
Likewise, if you are keeping paper copies, it would be a good idea to scan them electronically as well.
4. Hire a specialist to help
This is, of course, completely optional. That said. health insurance claims assistants exist to provide professional help for dealing with all your insurance records. Their services include filing and keeping track of an unlimited number of claims, reviewing medical bills for accuracy, and appealing rejected claims. If you have a particularly demanding need, these professionals can be an effective resource.
5. Online tool for people on Medicare
If you receive Medicare, it is important to know that there is an online tool for storing and accessing your personal information. You can use this resource to help you stay organized!
Finishing up the trial
As you're wrapping up participating in the study, you might have questions about what happens after the clinical trial ends. The end of the clinical trial does not mark the end of all communication between you and the research team. After your exit interviews with the research team to give you a chance to provide feedback on your experiences, you'll typically go back to having the standard care you were receiving before participating in the study. In some cases, you can continue on the study treatment - that and other details about what happens when the trial is over should be covered with you during the informed consent process before you enroll in the study.
The study staff will share details about the findings of the study with participants as soon as possible. If the results are positive, the treatment will become available to all patients as part of standard care.