When Grayson Schultz was 5 years old, he began to experience joint pain, swelling, fatigue, and a salmon-colored rash. After eight months of frequent doctor visits and a battery of tests, Grayson was diagnosed with systemic juvenile arthritis. The diagnoses didn’t stop there.
Over the next two decades, he was diagnosed with depression, anxiety, post-traumatic stress disorder (PTSD), postural orthostatic tachycardia syndrome (POTS), mast cell activation syndrome (MCAS), and hypermobility. He also began gender-affirming hormone therapy.

“I have a lot going on,” Schultz explains. “And on top of everything, managing my arthritis meds, while also navigating hormone replacement therapy is no easy task.” But Schultz feels extremely lucky to have a core group of competent doctors, including a rheumatologist, pain specialist, physical therapist, and primary care doctor, to oversee his complicated care.
“Being hypermobile means that I frequently dislocate different parts of my body, which typically requires an immediate trip to the ER or urgent care. However, being trans and queer, I choose to stay away from these places because the stigma and ignorance create a very hostile environment.”
Schultz isn't the only trans person to report experiencing hostility within a healthcare environment. A survey conducted by the Center for American Progress (CAP) found that 29% of transgender respondents said a doctor or other healthcare provider refused to see them because of their actual or perceived gender identity. They also found that 23% of transgender respondents said a doctor or healthcare provider had intentionally misgendered them or used the wrong name. Sadly, they also found that 29% of transgender survey takers had experienced unwanted physical contact from a doctor or healthcare provider (e.g. fondling, sexual assault)
Schultz admits that he encounters transphobia where he lives. “Many people lack experience with LGBTQ individuals and as a result, they have a fear of the unknown. And they don’t seem interested in understanding or learning. So, I prefer to stay home and deal with my medical issues on my own. I rely heavily on a team of doctors, who I completely trust.”

He adds, “It’s too bad that it has to be this way, but popping into an ER, explaining my life story, as well as my complex health history, to people who don’t understand me is just not worth it. The medical problem would have to be very serious for me to consider going. It’s sad, but it’s my reality.”
The Reality of LGBTQIA+ Diversity in Today’s Clinical Trials
Grayson Schultz’s health care experience is not uncommon. In fact, research shows that LGBTQIA+ individuals experience numerous health disparities and barriers linked to sexual orientation and gender identity. As a result, many members of the community choose to avoid health care or receive sub-standard care, due to societal stigma and discrimination by health care providers and organizations.
In addition to experiencing stigma, discrimination, and a denial of civil and human rights, many members of the LGBTQ community are omitted from clinical trials. And when it comes to testing new drugs, therapies, and devices, there is a huge lack of needed diversity in clinical trials. For example, a study of 243 clinical trials related to couples and sexual function after various medical treatments showed that 37 trials explicitly excluded people in same-sex relationships, according to the New England Journal of Medicine.
For the LGBTQ community, this limited diversity and inclusion stems from several issues, including mistrust; inaccessibility; inadequate engagement; lack of information about clinical trials and comfort with the research process; time and resource constraints associated with participation; and limited awareness of clinical trials.
According to a recent article written by Brandon Burch, Ph.D., director of scientific writing and regulatory affairs at Nuventra Pharma Sciences, “Diversity is important because there are many examples of drugs that behave differently from one population to another. Failing to understand these differences at the clinical trial stage could leave future patients at risk of a potentially avoidable safety issue or suboptimal efficacy. Put simply, a diverse and inclusive study population helps ensure an adequate understanding of how a drug works and how safe it is across the entire target population.”
He adds, “Ensuring accessibility, encouraging early engagement, and fostering communication and trust with patients benefits all stakeholders and shows a dedication to inclusivity and patient outcomes.”
Engagement, Trust, and Communication within the LGBTQIA+ Community
For Cedric Pulliam, Ph.D., a senior public health advisor at the Centers for Disease Control and Prevention — and a member of the black queer community — engagement, trust, and communication are essential to the entire clinical trial process. And community strategy groups are key to building this much-needed trust and engagement.
“Every clinical trial should begin with this type of strategy group,” he explains. “The purpose of the group is to help clinical trial developers and staff gather community input to develop protocols, which will ultimately ensure a successful and inclusive clinical study.”
He adds, “It’s all about the lived experience. Trial recruits need to see how the clinical trial is representative of their personal lifestyle. They need to be involved on the very first day—all the way until the very last day. It’s really about inclusion and giving everyone a voice. The idea of ‘can’t do this for us without us’ sums it up.”
When explaining the need to take trial participants into consideration, Pulliam offers an example of today’s LGBTQIA+ youth. “There is a very high rate of homelessness within the LGBTQ youth population,” he explains. “This high rate is typically due to family conflict, violence, neglect, abandonment, and a lack of acceptance. As a result, when these youth are included in clinical trials, the homeless scenario must be factored in to help develop successful recruitment practices, communication methods, logistics, and incentives for the trial.”
In addition to organizing a strategy group, health professionals who run clinical trials must have an understanding of the LGBTQIA+ community, including pronoun use, life experiences, and health practices.
“Many individuals avoid trials because health care providers don’t have the education or understanding needed to work with this population,” says Pulliam. “However, it means a great deal when doctors show they really care and want to learn. Doctors who give patients the time of day, as well as genuinely care about their lives, are a rare find.”
Nobody knows this more than Charis Hill. Hill, who is non-binary and trans, lives with axial spondyloarthritis (axSpA), a type of inflammatory arthritis, and Ehlers-Danlos syndrome, a connective tissue disorder, as well as anxiety, depression, and PTSD.

“Trust and communication are a very big part of my health journey,” explains Hill. “I need doctors who not only acknowledge the fact that I’m non-binary and trans, but also understand that I have a great deal of knowledge about my own unique health issues and respect me for having this insight.”
At the age of 13, Hill began to experience intense pain in their knees—with new symptoms appearing over time in their upper body, including cramping in the hips, extreme stiffness, lower back pain, and severe back spasms at night.
While Hill’s father had axial spondyloarthritis, their family believed axSpA only affected men, so Hill (who was assigned female at birth) didn't consider their symptoms to be a sign of something serious. In college, when Hill sought answers for their lower back pain, doctors did not suspect chronic disease. Instead, doctors only looked for signs of a sports injury, as Hill played college soccer, and blamed Hill's pain on being an athlete.
Years later, Hill sought answers again for symptoms that were only getting worse. When testing didn't show evidence of an underlying condition, doctors suggested they needed anti-anxiety medication and refused to explore other possibilities.
“As a result, I consistently felt like I had to prove that my pain was real, and not just in my head. I grew tired of the frequent gaslighting, the negative responses from doctors, and being told that my experiences were essentially fabricated. It was completely exhausting and made me realize that doctors do not know everything. I had to learn about the disease on my own and advocate for myself. Finally, at the age of 26, I was formally diagnosed with axial spondyloarthritis.”
Two years ago, Hill came out as non-binary. “I delayed this announcement because I knew it was going to be just one more thing I’d have to deal with. Being queer definitely adds another layer that must be navigated with medical professionals, due to the common lack of understanding. But I am learning that the patient/doctor relationship is 50/50.”
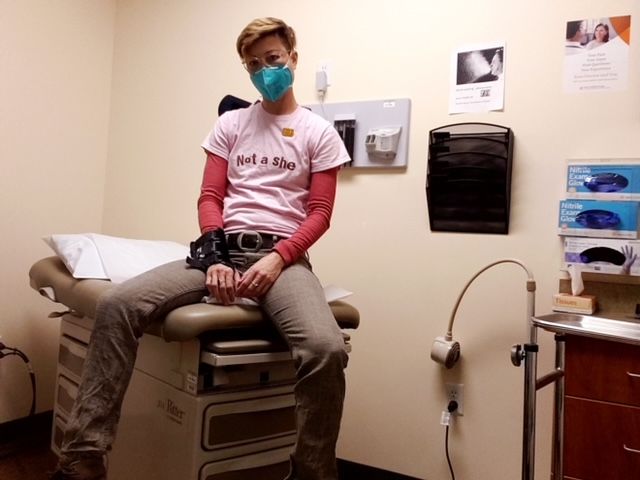
Hill adds, “The trust between me and my doctors needs to be a mutual trust. I want to trust my doctors— which includes a primary care doc, rheumatologist, OBGYN, psychiatrist, neurologist, and physiatrist—and they need to be able to trust me and know that I have done my homework. Many medical professionals seem to be less patient with chronic disease because they believe you can’t be fixed, but I refuse to settle for this point of view. And I shouldn’t have to.”
Increasing LGBTQ Inclusivity and Diversity in Clinical Trials
The shared experiences of Hill, Pulliam, and Schultz clearly highlight the need for change within the medical community. Efforts must be made to understand LGBTQIA+ health practices and improve access to quality healthcare for the entire LGBTQIA+ community. Additionally, it is vital to develop clinical trials and therapies within a diverse patient population.
“While this issue is acute in several clinical trial arenas, nowhere is it more so in the sometimes-underserved LGBTQIA+ population,” explains Katie Dry, a research physical therapist with the Military Health System via the Geneva Foundation. “Individuals might not seek healthcare because they do not feel like they are going to be treated equally. If you are not creating a culture that is inclusive and educated, people are not seeking care. It’s incumbent upon the clinical trial industry to build as broad a tent as possible.”
For trans folk, barriers to health care include include: financial barriers (lack of insurance and/or income), discrimination, lack of cultural understanding by providers, non-inclusive healthcare systems and socioeconomic barriers. While some of these barriers are faced by other marginalized groups, many are significantly magnified for transgender persons.
And there are numerous ways to build this broad tent. As part of their guidance on improving the diversity of clinical trials, designing inclusive trials, and overcoming recruiting barriers, the FDA provides a list of important and useful recommendations:
1. Make trial participation less burdensome for all participants
Reduce the frequency of study visits; consider flexibility in visits or electronic communication tools; and offer financial reimbursement for expenses associated with participation in clinical trials, such as travel and lodging.
2. Adopt enrollment and retention policies that enhance inclusiveness
Work with communities to address participant needs and involve patients, patient advocates, and caregivers in the design of clinical trial protocols; ensure clinical trial sites include locations with higher concentrations of minority patients; incorporate collaborative strategies for public outreach and education; hold recruiting events often and during convenient hours in trusted locations; and consider expanded access when appropriate. In addition, consider LGBTQ-specific issues, such as the high rate of homelessness among LGBTQ youth.
3. Engage patient advocacy groups early for suggestions on trial design
These groups are extremely helpful in reaching the right participants because they serve as a direct connection to the patient population.
4. Consider exactly what the study is intended to show and how the study can be designed to ensure the intended target population is adequately assessed.
5. Recognize and remove any barriers to study participation
Examples include accessibility; language; confidentiality concerns; cultural humility training for all staff; sexual orientation and gender identity (SO/GI) inclusive intake forms; inclusive strategies for public outreach and education; financial reimbursements for all expenses associated with clinical trial participation; and LGBTQIA+ specific resources.
Resources for LGBTQIA+ health
This guide offers resources on lesbian, gay, bisexual and transgender (LGBT) health
Fenway Health advocates for and delivers innovative, equitable, accessible health care, supportive services, and transformative research and education. They center LGBTQIA+ people, BIPOC individuals, and other underserved communities to enable our local, national, and global neighbors to flourish.
LGBT HealthLink, a program of CenterLink, is a community-driven national network of experts and professionals enhancing LGBT health by reducing health disparities within our communities.
References
Safer, Joshua D et al. “Barriers to healthcare for transgender individuals.” Current opinion in endocrinology, diabetes, and obesity vol. 23,2 (2016): 168-71.




